| Active substance |
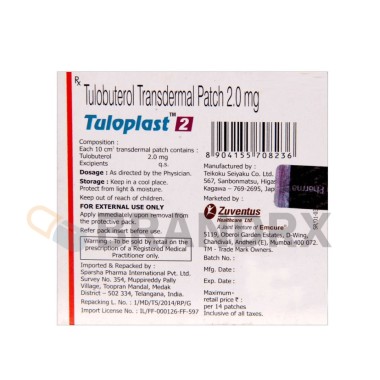
Tulobuterol |
| Also known as |
OPC-41061 |
| Blood pressure |
Generally has minimal effect on blood pressure |
| Chemical name |
N-[4-[(5R)-7-chloro-5-hydroxy-2-oxo-3-oxabicyclo[4.3.0]nona-1,6-dien-9-yl]pentyl]-1-methylbenzimidazole-2-carboxamide |
| Dosage (medical) |
Initial dose of 15 mg once daily, may be increased to a maximum of 60 mg daily based on serum sodium levels and clinical response |
| Dosage (sports) |
Not applicable as it is not typically used in sports |
| Effects |
Increases urine output without affecting the excretion of potassium and sodium, corrects hyponatremia |
| Formula |
C_26H_25ClN_2O_3 |
| Half-life |
Approximately 12 hours |
| Hepatotoxicity |
Can cause significant increases in liver enzymes, potential for serious liver injury |
| Lab Test |
Monitoring of serum sodium levels and liver function tests are necessary |
| Main action |
Vasopressin V2 receptor antagonist |
| Side effects |
Thirst, dry mouth, nausea, polyuria, hyperglycemia, raised liver enzymes |
| Storage conditions |
Store at room temperature away from moisture and heat |
| Strength |
2mg |
| Substance class |
Benzazepine derivative |
| Trade name |
Samsca |
| Use in sports |
None, as it is not known to enhance athletic performance |
| Water Retention |
Reduces water retention by promoting free water excretion |
| Manufacturer |
Zuventus Healthcare Ltd. |
| Packing |
14 patches/pack |