| Active substance |
Fexofenadine |
| Also known as |
Terfenadine metabolite |
| Blood pressure |
No significant effect |
| Chemical name |
(В±)-4-[1-hydroxy-4-[4-(hydroxydiphenylmethyl)-1-piperidinyl]butyl]-О±,О±-dimethyl benzeneacetic acid |
| Dosage (medical) |
60 mg twice daily or 180 mg once daily |
| Dosage (sports) |
Not applicable |
| Effects |
Reduces allergy symptoms such as sneezing, runny nose, itchy or watery eyes, and hives |
| Formula |
C32H39NO4 |
| Half-life |
14.4 hours |
| Hepatotoxicity |
Low |
| Lab Test |
Not specific |
| Main action |
H1 receptor antagonist |
| Side effects |
Headache, drowsiness, nausea, dizziness |
| Storage conditions |
Store at room temperature away from moisture |
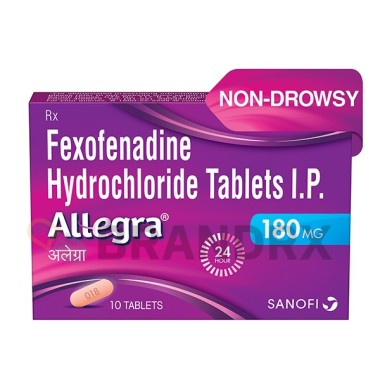
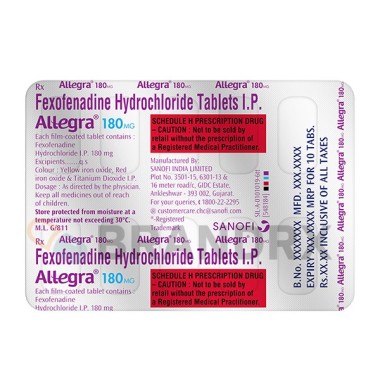
| Strength |
180mg |
| Substance class |
Antihistamine |
| Trade name |
Allegra, Telfast, Fexidine |
| Use in sports |
None |
| Water Retention |
No |
| Manufacturer |
Aventis Pharma Limited |
| Packing |
10 tabs/blister |